 Treatment Found To Reverse Hair Loss
Male pattern hair loss (MPHL) is a condition that affects as many as 50 percent of men by the age of fifty, but according to a new study, baldness may soon be a treatable condition.
In the study, one of each pair of twins was given 0.5 mg/day of the medication, the other receiving a placebo, with the results monitored over the course of a year. After both 6 and 12-month intervals, hair loss was found to not only have been halted in twins using dutasteride, but hair growth was actively improved. The rate of growth increased between the 6 and 12-month evaluations, while hair loss continued steadily in the placebo group. MPHL is thought to be genetically controlled. As identical twins share the same genetic code, the study provides particularly convincing evidence of the efficacy of dutasteride. Dutasteride (marketed as Avodart, Avidart, Avolve, Duagen, Dutas, Dutagen, Duprost) is a 5-alpha-reductase inhibitor, a drug which inhibits the conversion of testosterone into dihydrotestosterone (DHT). It is used to treat conditions caused by DHT, such as benign prostatic hyperplasia. Classification and Method of Action
Uses While dutasteride is officially approved, and most commonly used, to treat enlargement of the prostate gland, it may also be used to treat male pattern baldness. But, clinical trials for dutasteride as a hair loss drug were called off in late 2002. The reason the trials were called off is not outwardly known. However, industry sources speculate that Avodart would have been seen as too similar to Propecia to have proved profitable as a hair loss treatment. In December 2006, Avodart manufacturer GlaxoSmithKline embarked on a new Phase III, six month study in Korea to test the safety, tolerability and effectiveness of a once-daily dose of dutasteride (0.5mg) for the treatment of male pattern baldness in the vertex region of the scalp (types IIIv, IV and V on the Hamilton-Norwood scale).[1] The future impact that this study will have on the FDA's approval or disapproval of Avodart for the treatment of male pattern baldness in the United States is yet to be determined.
|
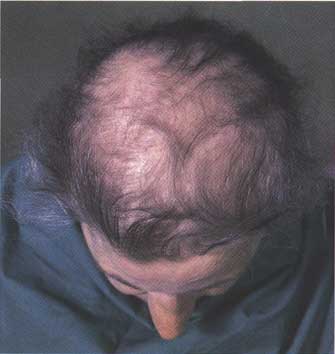 Using pairs of identical twins, the study assessed the efficacy of dutasteride, a particular enzyme inhibitor, as a means of reducing hair loss and encouraging new growth, finding it to be an effective means of combating baldness.
Using pairs of identical twins, the study assessed the efficacy of dutasteride, a particular enzyme inhibitor, as a means of reducing hair loss and encouraging new growth, finding it to be an effective means of combating baldness.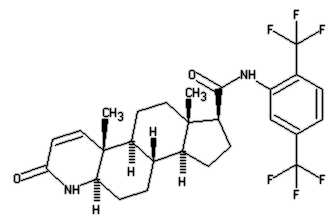 Dutasteride belongs to a class of drugs called 5-alpha-reductase inhibitors, which block the action of the 5-alpha-reductase enzymes that convert testosterone into dihydrotestosterone (DHT). Finasteride also belongs to this group. A clinical study done by GlaxoSmithKline, the EPICS trial, however, have not found dutasteride to be more effective than finasteride in treating BPH, even though it inhibits both isoforms of 5-alpha reductase, while finasteride inhibits only one.
Dutasteride belongs to a class of drugs called 5-alpha-reductase inhibitors, which block the action of the 5-alpha-reductase enzymes that convert testosterone into dihydrotestosterone (DHT). Finasteride also belongs to this group. A clinical study done by GlaxoSmithKline, the EPICS trial, however, have not found dutasteride to be more effective than finasteride in treating BPH, even though it inhibits both isoforms of 5-alpha reductase, while finasteride inhibits only one.